
Chemistry, 11.03.2020 00:24, MarishaTucker
Carbon monoxide gas reacts with hydrogen gas to form methanol via the following reaction: CO(g)+2H2(g)→CH3OH(g)CO(g)+2H2(g)→C H3OH(g) A 1.55 LL reaction vessel, initially at 305 KK, contains carbon monoxide gas at a partial pressure of 232 mmHgmmHg and hydrogen gas at a partial pressure of 389 mmHgmmHg . Identify the limiting reactant and determine the theoretical yeild of methonal in grams.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:00, haydjanggg6578
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2


Chemistry, 22.06.2019 19:10, asdfghhk9805
How does the atmosphere to make earth livable? check all that apply. causes the seasons contains oxygen provides warmth creates important nutrients blocks harmful energy from the sun plz like !
Answers: 2

Chemistry, 22.06.2019 21:00, cxttiemsp021
The rate constant for the reaction below is 6.2 x 10−5 mol l−1 s −1. if the initial concentration of a is 0.0500 m, what is its concentration after 115 s?
Answers: 1
Do you know the correct answer?
Carbon monoxide gas reacts with hydrogen gas to form methanol via the following reaction: CO(g)+2H2(...
Questions in other subjects:



Social Studies, 03.06.2021 08:20


Mathematics, 03.06.2021 08:20

Social Studies, 03.06.2021 08:20

Mathematics, 03.06.2021 08:20

History, 03.06.2021 08:20

English, 03.06.2021 08:20


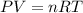 ..........(1)
..........(1)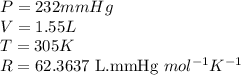
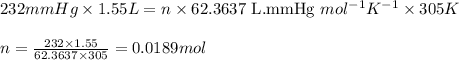
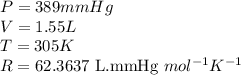
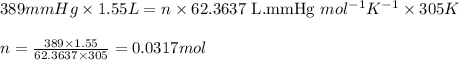
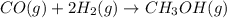
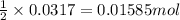 of carbon monoxide
of carbon monoxide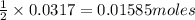 of methanol
of methanol




