The dissolution of PbI2(s) is represented above.

Chemistry, 10.03.2020 22:29, WhiteWinterRose
PbI2(s)⇄Pb2+(aq)+2I−(aq) Ksp=7×10−9
The dissolution of PbI2(s) is represented above.
(a) Write a mathematical expression that can be used to determine the value of S , the molar solubility of PbI2(s) .
(b) If PbI2(s) is dissolved in 1.0MNaI(aq), is the maximum possible concentration of Pb2+(aq) in the solution greater than, less than, or equal to the concentration of Pb2+(aq) in the solution in part (a) ? Explain.
Compound Ksp
PbCl2 2×10−5
PbI2 7×10−9
Pb(IO3)2 3×10−13
c) A table showing Ksp values for several lead compounds is given above. A saturated solution of which of the compounds has the greatest molar concentration of Pb2+(aq) ? Explain.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:30, freddhendrickss
When the speed of the bottle is 2 m/s, the average maximum height of the beanbag is m.
Answers: 2

Chemistry, 22.06.2019 16:00, yfnal3x
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2

Chemistry, 23.06.2019 07:50, dootdootkazoot
What is the significance sodium hydroxide and hydrochloric acid
Answers: 1
Do you know the correct answer?
PbI2(s)⇄Pb2+(aq)+2I−(aq) Ksp=7×10−9
The dissolution of PbI2(s) is represented above.
The dissolution of PbI2(s) is represented above.
Questions in other subjects:

History, 09.06.2020 18:57


Biology, 09.06.2020 18:57



History, 09.06.2020 18:57

Mathematics, 09.06.2020 18:57




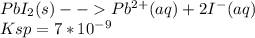
![K_s_p = [PB][I]^{2+}\\= s*2s\\s^3 = \frac{k_s_p}{4}](/tpl/images/0541/5945/0b3d5.png)
![s= \sqrt[3]{\frac{k_s_p}{4}}](/tpl/images/0541/5945/16063.png)
![s= \sqrt[3]{\frac{k_s_p}{4}}\\\\s= \sqrt[3]{\frac{7*10^-^9}{4}}](/tpl/images/0541/5945/27998.png)
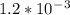 the maximum possible concentration of Pb²⁺ in the solution is always less than that in the solution in part (a). That is because of the common ion effect. The added iodide ion forces the position of equilibrium to shift to the left, reducing the concentration of Pb²⁺.
the maximum possible concentration of Pb²⁺ in the solution is always less than that in the solution in part (a). That is because of the common ion effect. The added iodide ion forces the position of equilibrium to shift to the left, reducing the concentration of Pb²⁺.![K_s_p = [Pb{^2^+}][X^-]^2](/tpl/images/0541/5945/b46ba.png)
![s =\sqrt [3]{\dfrac{K_{sp}}{4}}](/tpl/images/0541/5945/67f28.png)
![K_{sp} =\text{[Pb$^{2+}$][I$^{-}$]}^{2} = s\times (2s)^{2} = 4s^{3}\\s^{3} = \dfrac{K_{sp}}{4}\\\\s =\mathbf{ \sqrt [3]{\dfrac{K_{sp}}{4}}}\\\\\text{A mathematical expression you could use is }\mathbf{s =\sqrt [3]{\dfrac{K_{sp}}{4}}}](/tpl/images/0541/5945/67153.png)
![\begin{array}{rcl}\\s &=&\sqrt [3]{\dfrac{K_{\text{sp}}}{4}}\\\\s &=&\sqrt [3]{\dfrac{7 \times 10^{-9}}{4}}\\\\s &=&\sqrt [3]{1.75 \times 10^{-9}}\\\\&=& \mathbf{1.21 \times 10^{-3}} \textbf{ mol/L}\\\end{array}](/tpl/images/0541/5945/cc365.png)
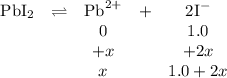
![K_{\text{sp}} =\text{[Pb$^{2+}$][I$^{-}$]}^{2} = s\times (1.0 + 2s)^{2} = 7 \times 10^{-9}\\s = \mathbf{7 \times 10^{-9}} \textbf{ mol/L}](/tpl/images/0541/5945/ddbab.png)
![K_{\text{sp}} =\text{[Pb$^{2+}$][X$^{-}$]}^{2}\\\\s = \text{[Pb$^{2+}$]} = \sqrt [3]{\dfrac{K_{\text{sp}}}{4}}](/tpl/images/0541/5945/770fe.png)




