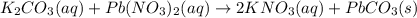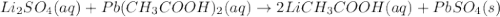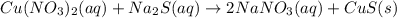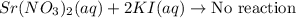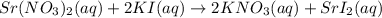Write a molecular equation for the precipitation reaction that occurs (if any) when the following solutions are mixed. If no reaction occurs, write NOREACTION.
Part A
potassium carbonate and lead(II) nitrate
Express your answer as a chemical equation. Enter NOREACTION if no reaction occurs. Identify all of the phases in your answer.
Part B
lithium sulfate and lead(II) acetate
Express your answer as a chemical equation. Enter NOREACTION if no reaction occurs. Identify all of the phases in your answer.
Part C
copper(II) nitrate and sodium sulfide
Express your answer as a chemical equation. Enter NOREACTION if no reaction occurs. Identify all of the phases in your answer.
Part D
strontium nitrate and potassium iodide
Express your answer as a chemical equation. Enter NOREACTION if no reaction occurs. Identify all of the phases in your answer.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:30, alexandroperez13
It takes 945.kj/mol to break a nitrogen-nitrogen triple bond. calculate the maximum wavelength of light for which a nitrogen-nitrogen triple bond could be broken by absorbing a single photon.
Answers: 3

Chemistry, 22.06.2019 06:00, rigobertogarza2
According to each substances heat of fusion, which of the items below requires more heat to be added per gram of substance to go from solid to liquid? silver sulfur water lead
Answers: 2

Chemistry, 22.06.2019 12:30, MrSavannahCat
Clyde and marilyn are riding a roller coaster. during which section(s) of the track is their potential energy converted to kinetic energy? a. from point b to point c only b. from point b to point d only c. from point a to point b only d. from point a to point b and from point c to point d
Answers: 1

Chemistry, 22.06.2019 17:00, calmicaela12s
Which statement is true about a catalyst? a: a catalyst decreases the rate of the reaction. b. a catalyst is consumed during a chemical reaction. c. a catalyst lowers the activation energy of a reaction. d. a catalyst increases the reactant concentration during a reaction.
Answers: 1
Do you know the correct answer?
Write a molecular equation for the precipitation reaction that occurs (if any) when the following so...
Questions in other subjects:

Physics, 10.09.2020 03:01


Mathematics, 10.09.2020 03:01

History, 10.09.2020 03:01

Mathematics, 10.09.2020 03:01

Mathematics, 10.09.2020 03:01

Mathematics, 10.09.2020 03:01

Geography, 10.09.2020 03:01

History, 10.09.2020 03:01

Chemistry, 10.09.2020 03:01