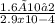Chemistry, 10.03.2020 17:11, sciencecreation87
In an analysis of the following reaction at a certain temperature, Br2(g) + Cl2(g) ⇌ 2BrCl(g)the equilibrium concentrations were found to be [Br2] = 4.5 ×10−3 M, [Cl2] = 2.6 ×10−2 M, and [BrCl] = 1.6 ×10−2 M. Write the equilibrium expression, and calculate the equilibrium constant for this reaction at this temperature.

Answers: 1
Other questions on the subject: Chemistry



Do you know the correct answer?
In an analysis of the following reaction at a certain temperature, Br2(g) + Cl2(g) ⇌ 2BrCl(g)the equ...
Questions in other subjects:






Mathematics, 28.06.2019 14:20





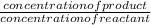
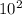
![[BrCl]^{2}](/tpl/images/0541/0381/fca7c.png)
![\frac{[BrCl[2}{[Br]2[Cl]2}](/tpl/images/0541/0381/5dca4.png) equation 1
equation 1![\frac{[1.6 ×10−2]}{[0.45 x 10^-2] [1.6 ×10−2]}](/tpl/images/0541/0381/7a675.png)