
50.0 mL solution of 0.160 M potassium alaninate ( H 2 NC 2 H 5 CO 2 K ) is titrated with 0.160 M HCl . The p K a values for the amino acid alanine are 2.344 ( p K a1 ) and 9.868 ( p K a2 ) , which correspond to the carboxylic acid and amino groups, respectively. a) how do you calculate the PH of the first equivalent and b) the second equivalent? please help

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 21.06.2019 19:00, 21brooklynmartin
Does the temperature affect the solubility of sugar and salt in water? if it does tell me like different temperatures with different solubilities so i can sketch down a graph
Answers: 2

Chemistry, 22.06.2019 21:30, Lindsay882
While in europe, if you drive 125 km per day, how much money would you spend on gas in one week if gas costs 1.10 euros per liter and your car’s gas mileage is 32.0 mi/gal? assume that 1 euro=1.26 dollars
Answers: 2

Chemistry, 22.06.2019 22:00, shaylasimonds587
The volume of an unknown substance in a sealed glass jar is 50 milliliters. the volume of the jar is 200 milliliters. which state of matter could the substance be?
Answers: 2
Do you know the correct answer?
50.0 mL solution of 0.160 M potassium alaninate ( H 2 NC 2 H 5 CO 2 K ) is titrated with 0.160 M HCl...
Questions in other subjects:


Social Studies, 22.10.2019 02:20

History, 22.10.2019 02:20

Social Studies, 22.10.2019 02:20


English, 22.10.2019 02:20



Chemistry, 22.10.2019 02:20


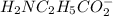

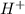 ------>
------> 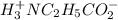
![[H_3}^+NC_2H_5CO^-_2]](/tpl/images/0540/7336/f75f8.png) =
= 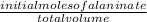
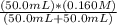
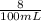
![[H^+]](/tpl/images/0540/7336/07acb.png) =
= ![\sqrt{\frac{K_{a1}K_{a2}{[H_3}^+NC_2H_5CO^-_2]+K_{a1}K_w}{ K_{a1}{[H_3}^+NC_2H_5CO^-_2] } }](/tpl/images/0540/7336/a0e17.png)
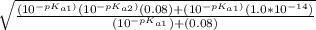

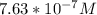
![-log[7.63*10^{-7}]](/tpl/images/0540/7336/a344f.png)
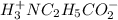
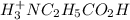
![[H^+_3NC_2H_5CO_2H]](/tpl/images/0540/7336/4ff0a.png) =
= 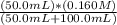
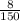
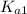 =
= ![\frac{[H^+] [H^+_3NC_2H_5CO^-_2]}{[H^+_3NC_2H_5CO_2H]}](/tpl/images/0540/7336/e2bce.png)
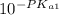 =
= 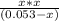
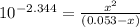
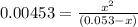
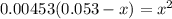
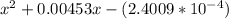
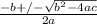
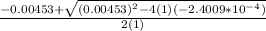 OR
OR 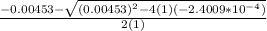
![[H^+]=[H_3^+NC_2H_5CO^-_2]= 0.0134 M](/tpl/images/0540/7336/73fad.png)
![-log[H^+]](/tpl/images/0540/7336/cbdd4.png)
![-log[0.0134]](/tpl/images/0540/7336/6b2f8.png)




