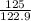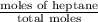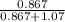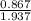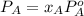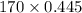Chemistry, 10.03.2020 07:25, starfox5454
At a certain temperature the vapor pressure of pure heptane (C_7 H_16) is measured to be 170. torr. Suppose a solution is prepared by mixing 86.7 g of heptane and 125. g of acetyl bromide (CH_3 COBr). Calculate the partial pressure of heptane vapor above this solution. Round your answer to 3 significant digits. Note for advanced students: you may assume the solution is ideal.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:20, ayoismeisalex
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 17:20, phanuel642
The small bags of silica gel you often see in a new shoe box are placed there to control humidity. despite its name, silica gel is a solid. it is a chemically inert, highly porous, amorphous form of sio2. because water vapor readily adsorbs onto the surface of silica gel, it acts as a desiccant. despite not knowing mechanistic details of the adsorption of water onto silica gel, from the information provided you should be able to make an educated guess about the thermodynamic characteristics of the process. predict the signs for δg, δh, and δs for the adsorption of water.
Answers: 2


Chemistry, 23.06.2019 00:40, joe7977
To prevent the presence of air, noble gases are placed over highly reactive chemicals to act as inert "blanketing" gases. a chemical engineer places a mixture of noble gases consisting of 4.37 g of he, 13.36 g of ne, and 36.65 g of kr in a piston-cylinder assembly at stp. calculate the partial pressure in torr of kr.
Answers: 1
Do you know the correct answer?
At a certain temperature the vapor pressure of pure heptane (C_7 H_16) is measured to be 170. torr....
Questions in other subjects:






Health, 08.02.2021 17:20

Mathematics, 08.02.2021 17:20

Mathematics, 08.02.2021 17:20

English, 08.02.2021 17:20


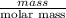
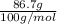
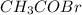 are calculated as follows.
are calculated as follows.