
Chemistry, 10.03.2020 06:00, TrueKing184
The equilibrium constant, Kc , for the decomposition of COBr2 COBr2(g) ↔ CO(g) + Br2(g) is 0.190. What is Kc for the following reaction? 2CO(g) + 2Br2(g) ↔ 2COBr2(g)

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, markmlg122
Two things that biomedical has invented or innvated
Answers: 1

Chemistry, 22.06.2019 05:50, zaleemawhite
Significant figures are digits read directly from the measuring instrument plus one more digit, which is __ by the observer.
Answers: 2

Chemistry, 22.06.2019 16:00, jrocklove7825
About 3% of the water on earth is freshest. only about 40% of that freshwater is available for human use. why is so much freshwater unavailable for human use?
Answers: 2

Chemistry, 22.06.2019 23:50, josie311251
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3
Do you know the correct answer?
The equilibrium constant, Kc , for the decomposition of COBr2 COBr2(g) ↔ CO(g) + Br2(g) is 0.190. Wh...
Questions in other subjects:


English, 30.09.2019 18:20

Mathematics, 30.09.2019 18:20

Health, 30.09.2019 18:20

Mathematics, 30.09.2019 18:20




History, 30.09.2019 18:20



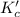 for the reaction.
for the reaction.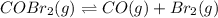

![K_c=\frac{[CO][Br_2]}{[COBr_2]}](/tpl/images/0540/1501/c4707.png) ..[1]
..[1]![K_c'=\frac{[COBr_2]^2}{[CO]^2[Br]^2}](/tpl/images/0540/1501/4595a.png)
 ( from [1])
( from [1])




