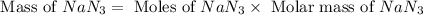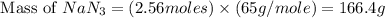The air bags in cars are inflated when a collision triggers the explosive, highly exothermic decomposition of sodium azide (NaN3): 2NaN3(s) → 2Na(s) + 3N2(g) The passenger-side air bag in a typical car must fill a space approximately four times as large as the driver-side bag to be effective. Calculate the mass of sodium azide required to fill a 113-L air bag. Assume the pressure in the car is 1.00 atm and the temperature of N2 produced is 85°C.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:50, vanessa051266
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 12:00, daytonalive83481
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 16:00, bbrogle5154
If 15 drops of ethanol from a medical dropper weight 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? the density of ethanol is 0.80g/ml
Answers: 1

Chemistry, 22.06.2019 23:00, poolwaterisgross
How does the value of the equilibrium constant show that a reaction reaches equilibrium very quickly? (a) the equilibrium constant is large. (b) the equilibrium constant is small. (c) the equilibrium constant is zero. (d) the value of the equilibrium constant does not show how quickly a reaction comes to equilibrium.
Answers: 1
Do you know the correct answer?
The air bags in cars are inflated when a collision triggers the explosive, highly exothermic decompo...
Questions in other subjects:





Chemistry, 14.09.2019 06:30



Computers and Technology, 14.09.2019 06:30



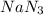 required is, 166.4 grams.
required is, 166.4 grams.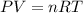
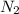 gas = 1.00 atm
gas = 1.00 atm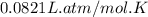
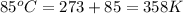
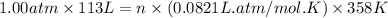
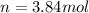
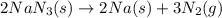
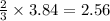 moles of
moles of