
Chemistry, 10.03.2020 04:52, Katie123amazing
Calculate ΔH∘f for NO(g) at 435 K, assuming that the heat capacities of reactants and products are constant over the temperature interval at their values at 298.15 K. Molar heat capacities of NO(g), N2(g), and O2(g) at 298.15 K are 29.86, 29.13, and 29.38 J⋅K−1⋅mol−1. The standard enthalpy of formation of NO(g) is 91.3 kJ⋅mol−1 at 298.15 K.

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 09:00, yogibear5806
Look at the spectrums of a star moving towards earth and a motionless star. which of these is a correct inference that can be draw from the observation of the two spectrums? (2 points) the spectrum of a motionless star is difficult to be viewed separately using oridinary telescopes. the spectrum of a motionless star is identical to the spectrum of a star which moves towards earth. the spectrum of a star shifts towards the red region when the star moves towards earth. the spectrum of a star shifts towards the blue region when the star moves towards earth.
Answers: 2

Chemistry, 22.06.2019 21:30, sierradanielle9280
In science class richard learns that a substance has a boiling point of 230 fahrenheit his teacher ask him to convert this temperature to degrees celsius what is the boiling point of his substance in degrees celsius
Answers: 3
Do you know the correct answer?
Calculate ΔH∘f for NO(g) at 435 K, assuming that the heat capacities of reactants and products are c...
Questions in other subjects:

Mathematics, 26.09.2021 02:50

Mathematics, 26.09.2021 02:50

Chemistry, 26.09.2021 02:50


Chemistry, 26.09.2021 02:50


Mathematics, 26.09.2021 02:50




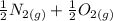 ------>
------>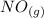
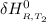 =
= 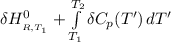
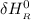 = enthalpy of reaction
= enthalpy of reaction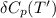 = the difference in the heat capacities of the products and the reactants.
= the difference in the heat capacities of the products and the reactants.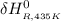 =
= 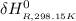

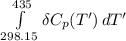
![1(91300 J.mol^{-1} ) +\int\limits^{435}_{298.15} [{(29.86)-\frac{1}{2}(29.38)-\frac{1}{2}29.13}]J.K^{-1}.mol^{-1} \, dT'](/tpl/images/0540/0534/3b971.png)





