
Chemistry, 10.03.2020 04:50, MathChic68
The following reaction was performed in a sealed vessel at 791 ∘C : H2(g)+I2(g)⇌2HI(g) Initially, only H2 and I2 were present at concentrations of [H2]=3.10M and [I2]=2.50M . The equilibrium concentration of I2 is 0.0800 M . What is the equilibrium constant, Kc, for the reaction at this temperature?

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 07:30, danielahumajova6
How do you interpret a chromagram for what mixtures contain?
Answers: 1
Do you know the correct answer?
The following reaction was performed in a sealed vessel at 791 ∘C : H2(g)+I2(g)⇌2HI(g) Initially, on...
Questions in other subjects:

Mathematics, 30.01.2020 08:50

Health, 30.01.2020 08:50

Chemistry, 30.01.2020 08:50

Arts, 30.01.2020 08:50





Mathematics, 30.01.2020 08:50

Business, 30.01.2020 08:50


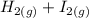 ⇌
⇌ 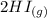
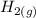

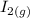 ⇌
⇌ ![K_c = \frac{[HI]^2}{[H_2][I_2]}](/tpl/images/0540/0427/6b81e.png)
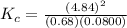
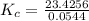
 430.62
430.62 ≅ 431
≅ 431 



