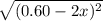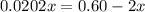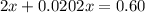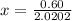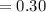Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 13:30, Unstinct
Lithium diisopropylamide [(ch3)2ch]2nli, referred to as lda, enjoys many uses as a strong base in synthetic organic chemistry. it is customarily prepared by the reaction of diisopropylamine [(ch3)2ch]2nh with butyllithium. draw the products of the reactions in the appropriate boxes and select the acid, base, conjugate acid, and conjugate base. be sure to answer all parts.
Answers: 2


Do you know the correct answer?
At 2000°C, the equilibrium constant for the reaction below is Kc = 4.10 ´ 10–4 . If 0.600 moles of N...
Questions in other subjects:

Mathematics, 08.08.2019 03:10




Mathematics, 08.08.2019 03:10






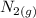 is going to be 0.30M
is going to be 0.30M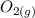 ⇄ 2
⇄ 2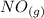
![K_{c}=\frac{[products]^{stoichiometric coefficient} }{[reactants]^{stoichiometric coefficient} }](/tpl/images/0539/5556/af056.png)
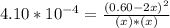
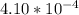 =
= 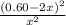
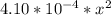 =
= 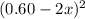
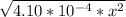 =
=