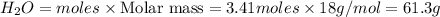Chemistry, 09.03.2020 23:57, jaynissiagreen16
The Ostwald process is used commercially to produce nitric acid, which is, in turn, used in many modern chemical processes. In the first step of the Ostwald process, ammonia is reacted with oxygen gas to produce nitric oxide and water. What is the maximum mass of H 2 O that can be produced by combining 91.0 g of each reactant? 4 NH 3 ( g ) + 5 O 2 ( g ) ⟶ 4 NO ( g ) + 6 H 2 O ( g )

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 17:00, davisnaziyahovz5sk
The arrangement of particles is most ordered in a sample of
Answers: 1


Chemistry, 23.06.2019 07:00, kotetravels10
0.88 moles of n2o5 (g) was placed in a sealed 1.00 l vessel. calculate the equilibrium concentration of n2o5. no2, and o2 and the equilibrium constant after equilibrium has been reached by 65.0% of the n2o5 decomposing.
Answers: 1

Chemistry, 23.06.2019 08:30, elijah4723
If you had to research a particular disease or area of concern in veterinary medicine and science, which one would you choose? why?
Answers: 1
Do you know the correct answer?
The Ostwald process is used commercially to produce nitric acid, which is, in turn, used in many mod...
Questions in other subjects:


Mathematics, 02.07.2020 23:01


History, 02.07.2020 23:01

Mathematics, 02.07.2020 23:01

Mathematics, 02.07.2020 23:01

Mathematics, 02.07.2020 23:01

Biology, 02.07.2020 23:01


Mathematics, 02.07.2020 23:01


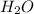 will be produced from the given masses of both reactants.
will be produced from the given masses of both reactants.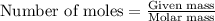
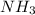
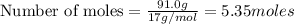
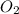
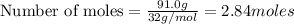
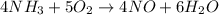
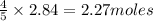 of
of 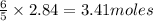 of
of