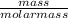What causes gas particles to collide with the walls of their containers?

Chemistry, 09.03.2020 06:46, stevencardona12mejia
Question 1.
What causes gas particles to collide with the walls of their containers?
A : Their volume
B : Their constant motion
C : Their pressure
D : Their small size
Question 2.
A balloon is filled with 5 grams of Helium gas. If the balloon takes up 10 mL of space and has a pressure of 20 mm Hg, which gas law needs to be used to find the temperature inside the balloon?
A: Charles law
B: Boyle’s law
C: Ideal gas law
D: Combined gas law

Answers: 2
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 11:50, hadwell34
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 22.06.2019 15:20, shanyeah
Water is initially present in a state where its molecules are far apart. during a change of state, its molecules slow down. which change of state has most likely taken place? from a gas to a liquid from a liquid to a gas from a solid to a liquid from a gas to a plasma
Answers: 1
Do you know the correct answer?
Question 1.
What causes gas particles to collide with the walls of their containers?
What causes gas particles to collide with the walls of their containers?
Questions in other subjects:


Mathematics, 15.07.2020 15:01


Mathematics, 15.07.2020 15:01

Biology, 15.07.2020 15:01

Mathematics, 15.07.2020 15:01

Social Studies, 15.07.2020 15:01


Mathematics, 15.07.2020 15:01