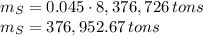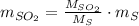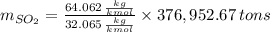Chemistry, 07.03.2020 05:01, qudoniselmore0
In 1986 an electrical power plant in Taylorsville, Georgia, burned 8,376,726 \rm tons of coal, a national record at that time.
Assuming that the coal was 86.0 \% carbon by mass and that combustion was complete, calculate the number of tons of carbon dioxide produced by the plant during the year.
Assuming that the coal was 4.50 \% sulfur by mass and that combustion was complete, calculate the number of tons of sulfur dioxide produced by the plant during the year.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:30, umimgoingtofail
Motivation cannot be developed with practice; a person either possesses it or they do not.
Answers: 1


Chemistry, 22.06.2019 09:30, janetexcoelho
What does the mass of 0.7891 mol of ferric oxide (fe2o3)
Answers: 1
Do you know the correct answer?
In 1986 an electrical power plant in Taylorsville, Georgia, burned 8,376,726 \rm tons of coal, a nat...
Questions in other subjects:


Biology, 03.02.2021 01:00






Physics, 03.02.2021 01:00



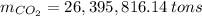 , b)
, b) 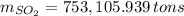
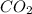 is produced by a mole of
is produced by a mole of 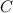 contained in coal. The yearly burnt carbon is:
contained in coal. The yearly burnt carbon is: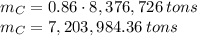
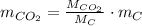
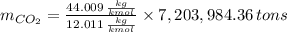
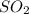 is produced by a mole of
is produced by a mole of 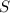 contained in coal.
contained in coal.