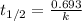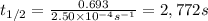Chemistry, 07.03.2020 04:45, Gghbhgy8716
The decomposition of N2O5 is described by the following equation. 2N2O5(g) → 4NO2(g) + O2(g) If the rate constant is 2.50 × 10−4 s−1, what is the half-life of this reaction?

Answers: 2
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 12:40, whitethunder05
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2

Chemistry, 22.06.2019 14:00, Killion2022
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1
Do you know the correct answer?
The decomposition of N2O5 is described by the following equation. 2N2O5(g) → 4NO2(g) + O2(g) If the...
Questions in other subjects:

Physics, 22.07.2021 07:00

Mathematics, 22.07.2021 07:00

English, 22.07.2021 07:00

English, 22.07.2021 07:00

English, 22.07.2021 07:00


Physics, 22.07.2021 07:00




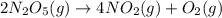
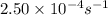
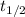 ) and rate constant (k)are related by :
) and rate constant (k)are related by :