
Chemistry, 07.03.2020 04:52, Messick127
A sample of dangerously reactive fluorine gas is contained in a stainless steel container which has a 31 L capacity at a pressure of 160 pounds per square inch and a temperature of 26.0◦C. What mass of fluorine gas sample is in the stainless steel container? 1. 10.3098 g 2. 522.362 g 3. 6.87318 g 4. 27.4927 g 5. 18.3285 g

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, armahoney8566
Gymnast always perform on padded mats. how does the mats protect the gymnast
Answers: 2


Chemistry, 22.06.2019 19:30, simihehe
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2

Chemistry, 22.06.2019 20:40, ohgeezy
Select the correct value for the indicated bond angle in each of the compounds. o−o−oo−o−o angle of o3 90° 109.5° < 109.5° 120° < 120° 180° f−b−ff−b−f angle of bf3 180° < 109.5° < 120° 120° 109.5° 90° f−o−ff−o−f angle of of2 < 120° 120° 90° 109.5° 180° < 109.5° cl−be−clcl−be−cl angle of becl2 90° 109.5° 180° 120° < 109.5° < 120° f−p−ff−p−f angle of pf3 90° 109.5° < 109.5° 180° 120° < 120° h−c−hh−c−h angle of ch4 90° < 109.5° 180° 120° < 120° 109.5°
Answers: 1
Do you know the correct answer?
A sample of dangerously reactive fluorine gas is contained in a stainless steel container which has...
Questions in other subjects:

Mathematics, 23.02.2021 20:50

Mathematics, 23.02.2021 20:50






Health, 23.02.2021 20:50

History, 23.02.2021 20:50


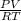 =
= 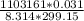 = 13.75 moles of fluorine
= 13.75 moles of fluorine



