Chemistry, 07.03.2020 00:44, vjackie101ov3kju
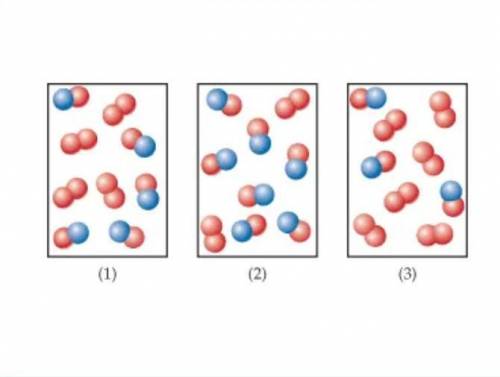
The following diagrams represent mixtures of NO(g) and O2(g). These two substances react as follows: 2NO(g)+O2(g)→2NO2(g) It has been determined experimentally that the rate is second order in NO and first order in

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:30, joejoefofana
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀ pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4. 0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3



Chemistry, 22.06.2019 23:00, NewKidnewlessons
What is the energy in joules of a mole of photons associated with visible light of wavelength 486 nm?
Answers: 3
Do you know the correct answer?
The following diagrams represent mixtures of NO(g) and O2(g). These two substances react as follows:...
Questions in other subjects:






Chemistry, 18.05.2021 19:10



Chemistry, 18.05.2021 19:10

History, 18.05.2021 19:10