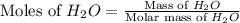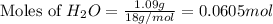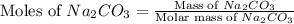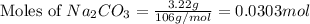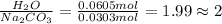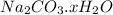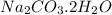Chemistry, 07.03.2020 00:25, ginalopez567
Dumbledore decides to gives a surprise demonstration. He starts with a hydrate of Na2CO3 which has a mass of 4.31 g before heating. After he heats it he finds the mass of the anhydrous compound is found to be 3.22 g. He asks everyone in class to determine the integer x in the hydrate: Na2CO3·xH2O; you should do this also. Round your answer to the nearest integer.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 23:30, sanociahnoel
The density of the solid phase of a substance is 0.90 g/cm3 and the density of the liquid phase is 1.0 g/cm3. a large increase in pressure will a. lower the freezing point b. raise the freezing point c. lower the boiling point d. raise the triple point e. lower the triple point
Answers: 1

Chemistry, 23.06.2019 07:00, asims13
The following transition occurs at a molecular level for a substance. what transition corresponds to this change in microscopic structure? the carbon dioxide molecules on the left are in a regular, tightly packed pattern. after heating, it becomes much lower density. a. melting b. boiling c. sublimation d. freezing
Answers: 1
Do you know the correct answer?
Dumbledore decides to gives a surprise demonstration. He starts with a hydrate of Na2CO3 which has a...
Questions in other subjects:




Chemistry, 04.08.2019 02:00



Physics, 04.08.2019 02:00

History, 04.08.2019 02:00

History, 04.08.2019 02:00

Business, 04.08.2019 02:00


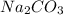 = 4.31 g
= 4.31 g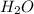 and
and