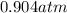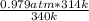Chemistry, 06.03.2020 23:49, RoyalGurl01
A sealed container holding 0.0255 L of an ideal gas at 0.979 atm and 67 ∘ C is placed into a refrigerator and cooled to 41 ∘ C with no change in volume. Calculate the final pressure of the gas. P = atm

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 22:30, darceline1574
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3


Chemistry, 23.06.2019 02:00, jacckiie5176
Which of these is a density dependent factor? a. epidemic b. earthquake c. drought d. hurricane
Answers: 2
Do you know the correct answer?
A sealed container holding 0.0255 L of an ideal gas at 0.979 atm and 67 ∘ C is placed into a refrige...
Questions in other subjects:



Mathematics, 09.11.2020 19:50


History, 09.11.2020 19:50

Mathematics, 09.11.2020 19:50

Social Studies, 09.11.2020 19:50

Biology, 09.11.2020 19:50