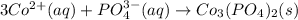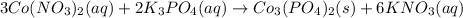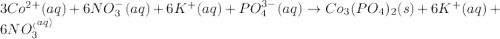Chemistry, 06.03.2020 23:39, ladypink94
When aqueous solutions of cobalt(II) nitrate and potassium phosphate are combined, solid cobalt(II) phosphate and a solution of potassium nitrate are formed. The net ionic equation for this reaction is :

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:00, emilyproce
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1


Chemistry, 22.06.2019 21:30, kawaiiblurainbow
Which of the following changes will decrease the total amount of gaseous solute able to be dissolved in a liter of liquid water? (2 points) decreasing temperature decreasing pressure decreasing surface area decreasing solute concentration
Answers: 1
Do you know the correct answer?
When aqueous solutions of cobalt(II) nitrate and potassium phosphate are combined, solid cobalt(II)...
Questions in other subjects:


Mathematics, 13.08.2020 22:01







Mathematics, 13.08.2020 22:01