
Chemistry, 06.03.2020 23:26, lilquongohard
You carefully weigh out 16.00 g of CaCO3 powder and add it to 64.80 g of HCl solution. You notice bubbles as a reaction takes place. You then weigh the resulting solution and find that it has a mass of 74.24 g . The relevant equation is CaCO_3(s) + 2HCl(aq) rightarrow H_2O(l) + CO_2(g) + CaCl_2(aq) Assuming no other reactions take place, what mass of CO_2 was produced in this reaction?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:00, happy121906
Which position represents spring in the southern hemisphere? a) b) c) d)
Answers: 2

Chemistry, 22.06.2019 19:00, cindyroxana229
Nan element’s square on the periodic table, the number with the greatest numerical value represents the
Answers: 3

Chemistry, 23.06.2019 02:30, paulinahunl17
What type of energy conversion occurs when you place your feet near the fire place and they become warm
Answers: 1

Chemistry, 23.06.2019 05:00, andrwisawesome0
Match each term to its description. match term definition excess reactant a) reactant that can produce a lesser amount of the product limiting reactant b) amount of product predicted to be produced by the given reactants theoretical yield c) reactant that can produce more of the product
Answers: 3
Do you know the correct answer?
You carefully weigh out 16.00 g of CaCO3 powder and add it to 64.80 g of HCl solution. You notice bu...
Questions in other subjects:


Mathematics, 18.03.2021 02:30


History, 18.03.2021 02:30

Mathematics, 18.03.2021 02:30




English, 18.03.2021 02:30

History, 18.03.2021 02:30


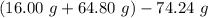
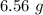
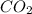 produced in this reaction was 6.56 grams
produced in this reaction was 6.56 grams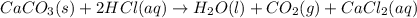
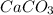 + mass of
+ mass of 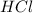 = 16.00 + 64.80 = 80.80 g
= 16.00 + 64.80 = 80.80 g



