
Chemistry, 06.03.2020 16:58, ramireztony741
Using the equations 2 Sr(s) + O₂ (g) → 2 SrO (s) ∆H° = -1184 kJ/mol SrO (s) + CO₂ (g) → SrCO₃ (s) ∆H° = -234 kJ/mol CO₂ (g) → C(s) + O₂ (g) ∆H° = 394 kJ/mol Determine the enthalpy for the reaction 2 SrCO₃ (s) → 2 Sr (s) + 2 C(s) + 3 O₂ (g).

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 16:00, matt16913
Which of the following is the correct definition of chemical energy? a. energy an object has because of its motion or position b. energy resulting from the flow of charged particles, such as electrons or ions c. energy produced from the splitting of atoms d. energy stored in chemical bonds of molecules
Answers: 1

Chemistry, 22.06.2019 22:30, kristen17diaz
How many valence electrons are in atom of radon?
Answers: 1
Do you know the correct answer?
Using the equations 2 Sr(s) + O₂ (g) → 2 SrO (s) ∆H° = -1184 kJ/mol SrO (s) + CO₂ (g) → SrCO₃ (s) ∆H...
Questions in other subjects:

Mathematics, 22.10.2021 14:00

Mathematics, 22.10.2021 14:00


English, 22.10.2021 14:00



Mathematics, 22.10.2021 14:00

Chemistry, 22.10.2021 14:00

English, 22.10.2021 14:00

Physics, 22.10.2021 14:00


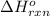 for the reaction is 72 kJ.
for the reaction is 72 kJ.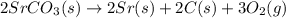
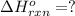
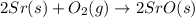
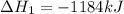
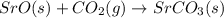
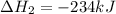 ( × 2)
( × 2)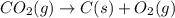
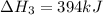 ( × 2)
( × 2)![\Delta H^o_{rxn}=[1\times (\Delta H_1)]+[2\times (-\Delta H_2)]+[2\times (\Delta H_3)]](/tpl/images/0535/5885/feb85.png)
![\Delta H^o_{rxn}=[(1\times (-1184))+(2\times -(-234))+(2\times (394))]=72kJ](/tpl/images/0535/5885/d98fd.png)




