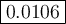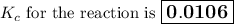Calculate the value of the equilibrium constant, K c , for the reaction Q ( g ) + X ( g ) − ⇀ ↽ − 2 M ( g ) + N ( g ) given that M ( g ) − ⇀ ↽ − Z ( g ) K c 1 = 3.15 6 R ( g ) − ⇀ ↽ − 2 N ( g ) + 4 Z ( g ) K c 2 = 0.509 3 X ( g ) + 3 Q ( g ) − ⇀ ↽ − 9 R ( g ) K c 3 = 12.5

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, deidaraXneji
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 3


Chemistry, 22.06.2019 09:20, pandaman632
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1
Do you know the correct answer?
Calculate the value of the equilibrium constant, K c , for the reaction Q ( g ) + X ( g ) − ⇀ ↽ − 2...
Questions in other subjects:


English, 06.12.2019 04:31


History, 06.12.2019 04:31

Social Studies, 06.12.2019 04:31

Biology, 06.12.2019 04:31


Mathematics, 06.12.2019 04:31

Social Studies, 06.12.2019 04:31

Social Studies, 06.12.2019 04:31