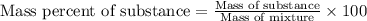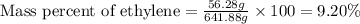Ethylene () is the starting point for a wide array of industrial chemical syntheses. For example, worldwide about of polyethylene are made from ethylene each year, for use in everything from household plumbing to artificial joints. Natural sources of ethylene are entirely inadequate to meet world demand, so ethane () from natural gas is "cracked" in refineries at high temperature in a kinetically complex reaction that produces ethylene gas and hydrogen gas. Suppose an engineer studying ethane cracking fills a reaction tank with of ethane gas and raises the temperature to . He believes at this temperature. Calculate the percent by mass of ethylene the engineer expects to find in the equilibrium gas mixture. Round your answer to significant digits. Note for advanced students: the engineer may be mistaken about the correct value of , and the mass percent of ethylene you calculate may not be what he actually observes.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:30, UaRemomGAY
If anyone would be able to me out with these three questions it would be these are from the chem 2202 course.
Answers: 3

Chemistry, 23.06.2019 01:00, aliviadushane
If a straight-chain hydrocarbon is a gas at room temperature, how many carbon atoms will it have? a. 6 carbon atoms b. 12 carbon atoms c. 24 carbon atoms d. 3 carbon atoms
Answers: 1

Do you know the correct answer?
Ethylene () is the starting point for a wide array of industrial chemical syntheses. For example, wo...
Questions in other subjects:





English, 25.09.2021 14:00


Mathematics, 25.09.2021 14:00

English, 25.09.2021 14:00

Physics, 25.09.2021 14:00


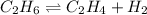
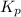 for above equation follows:
for above equation follows: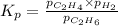
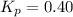
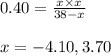
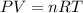 ..........(1)
..........(1)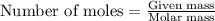 .....(2)
.....(2)![P=34.3atm\\V=30.0L\\R=0.0821\text{ L atm }mol^{-1}K^{-1}\\T=400^oC=[400+273]=673K](/tpl/images/0534/8160/3194c.png)

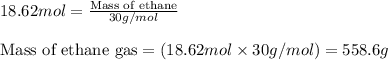
![P=3.70atm\\V=30.0L\\R=0.0821\text{ L atm }mol^{-1}K^{-1}\\T=400^oC=[400+273]=673K](/tpl/images/0534/8160/4b69b.png)