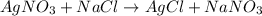Chemistry, 05.03.2020 00:22, tytybruce2
On a balance, you have beakers of AgNO3AgNO3 solution and NaClNaCl solution. When mixed, they will form AgCl(s)AgCl(s). What will happen to the mass of the new mixture?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, pinkycupcakes3oxbqhx
200. ml of 3.00 m nacl solution is diluted to a final volume of 500. ml. what is the molarity of the final solution?
Answers: 2

Chemistry, 22.06.2019 06:30, AleciaCassidy
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1


Chemistry, 23.06.2019 01:30, emfranco1
Which of the following statements is true about energy quantization at the atomic level? electrons in the outermost orbits are the most stable. electrons in all the orbits around the nucleus have the same amount of energy. electrons in the orbit closest to the nucleus have the least amount of energy. electrons absorb or release the same amount of energy independent of the energy levels.
Answers: 1
Do you know the correct answer?
On a balance, you have beakers of AgNO3AgNO3 solution and NaClNaCl solution. When mixed, they will f...
Questions in other subjects:

Biology, 21.07.2019 21:10


Mathematics, 21.07.2019 21:10

Biology, 21.07.2019 21:10

Mathematics, 21.07.2019 21:10

Chemistry, 21.07.2019 21:10

Mathematics, 21.07.2019 21:10

History, 21.07.2019 21:10


Mathematics, 21.07.2019 21:10


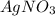 is added to NaCl then the compound formed will have same mass as that of reactants.
is added to NaCl then the compound formed will have same mass as that of reactants.