
Chemistry, 04.03.2020 23:26, TheOneandOnly003
A compound is 92.2% Carbon and 7.76% Hydrogen. The formula mass of the compound is 78.1 g. Calculate the empirical formula and molecular formula of the compound.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:00, leahstubbs
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 22.06.2019 23:00, catdog5225
What is formed when amino acids form long chains or polymerize
Answers: 1

Chemistry, 23.06.2019 07:00, Teddybearnerd
The image compares the arrangement of electrons in two different neutral atoms. a figure labeled atom q has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has six black spheres. to the left of this figure is another figure labeled atom p. atom p has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has seven black spheres. which of the following best explains the position of the two atoms in the periodic table?
Answers: 2

Chemistry, 23.06.2019 08:00, mackaylabarnes22
Ineed this awnser fast select the correct answer. this chemical equation represents the burning of methane, but the equation is incomplete. what is the missing coefficient in both the reactants and the products? ch4 + → co2 + a. 0 b. 1c. 2d. 3 e. 4
Answers: 3
Do you know the correct answer?
A compound is 92.2% Carbon and 7.76% Hydrogen. The formula mass of the compound is 78.1 g. Calculate...
Questions in other subjects:





Biology, 15.04.2021 16:20


Mathematics, 15.04.2021 16:20

Mathematics, 15.04.2021 16:20



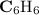
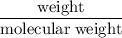
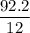
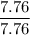
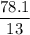
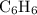 .
.



