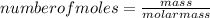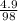Chemistry, 04.03.2020 23:40, malachilaurenc
Ammonium phosphate ((NH4)3PO4) is an important ingredient in many fertilizers. It can be made by reacting phosphoric acid (H3PO4) with ammonia (NH3).
What mass of ammonium phosphate is produced by the reaction of 4.9 g phosphoric acid?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:00, hammackkatelyn60
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3

Chemistry, 23.06.2019 04:20, monifaWilson
The graph shows one consequence of urban sprawl. how did urban sprawl contribute to the change in biodiversity
Answers: 2


Chemistry, 23.06.2019 06:30, ayoismeisjjjjuan
What is the chemical formula for a compound between li and br? libr li2br libr2 libr3
Answers: 1
Do you know the correct answer?
Ammonium phosphate ((NH4)3PO4) is an important ingredient in many fertilizers. It can be made by rea...
Questions in other subjects:


English, 10.02.2020 02:25


Mathematics, 10.02.2020 02:25


Mathematics, 10.02.2020 02:25

Arts, 10.02.2020 02:25

Mathematics, 10.02.2020 02:25

Mathematics, 10.02.2020 02:25

French, 10.02.2020 02:25


![3NH_3 +H_3PO_4 ----------- [{(NH_4)_3PO_4]](/tpl/images/0533/9487/9ce0d.png)