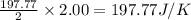Consider the reaction 2CO(g) + 2NO(g)2CO2(g) + N2(g) Using standard thermodynamic data at 298K, calculate the entropy change for the surroundings when 2.00 moles of CO(g) react at standard conditions. S°surroundings = J/K Are You Sure? Please check your answer for mistakes. Submit Answer

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, rebeccacruzz2017
Calculate - analysis of compound composed of iron and oxygen yields 174.86 of fe and 75.14g of o. what is the empirical formula for this compound?
Answers: 3



Chemistry, 23.06.2019 00:50, lakhanir2013
The chemical formula for emerald is be3al2(sio3)6.an emerald can be decided as
Answers: 3
Do you know the correct answer?
Consider the reaction 2CO(g) + 2NO(g)2CO2(g) + N2(g) Using standard thermodynamic data at 298K, calc...
Questions in other subjects:











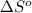 for the surrounding when given amount of CO gas is reacted is 197.77 J/K
for the surrounding when given amount of CO gas is reacted is 197.77 J/K![\Delta S^o_{rxn}=\sum [n\times \Delta S^o_{(product)}]-\sum [n\times \Delta S^o_{(reactant)}]](/tpl/images/0533/9520/52737.png)
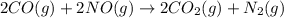
![\Delta S^o_{rxn}=[(2\times \Delta S^o_{(CO_2(g))})+(1\times \Delta S^o_{(N_2(g))})]-[(2\times \Delta S^o_{(CO(g))})+(2\times \Delta S^o_{(NO(g))})]](/tpl/images/0533/9520/8ee2e.png)

![\Delta S^o_{rxn}=[(2\times (213.74))+(1\times (191.61))]-[(2\times (197.67))+(2\times (210.76))]\\\\\Delta S^o_{rxn}=-197.77/K](/tpl/images/0533/9520/e8ddc.png)