
Chemistry, 03.03.2020 17:11, AgarioEdit
Calculate the amount of heat needed to melt 91.5g of solid benzene ( C6H6 ) and bring it to a temperature of 60.6°C . Round your answer to 3 significant digits. Also, be sure your answer contains a unit symbol.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:50, mrylenastewart
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3 -> a2cr(no3)3 + a3ag2so4
Answers: 1


Chemistry, 22.06.2019 09:00, kkmonsterhigh18
The diagram below shows a cell placed in a solution. a cell is shown placed inside a beaker. it is labeled cell. the solution inside the beaker is labeled 40% salt solution and the solution inside the cell is labeled 20% salt solution. only water is allowed to move in and out of the cell. what will most likely happen to the cell? it will expand as water moves out of it. it will shrink as water moves out of it. it will expand as water moves into it. it will shrink as water moves into it.
Answers: 2

Chemistry, 22.06.2019 13:00, wbrandi118
Using the thermodynamic information in the aleks data tab, calculate the standard reaction free energy of the following chemical reaction: →+p4o10s6h2ol4h3po4s round your answer to zero decimal places.
Answers: 3
Do you know the correct answer?
Calculate the amount of heat needed to melt 91.5g of solid benzene ( C6H6 ) and bring it to a temper...
Questions in other subjects:

Mathematics, 29.01.2020 02:06

English, 29.01.2020 02:06

Mathematics, 29.01.2020 02:06


History, 29.01.2020 02:06


History, 29.01.2020 02:06




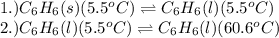
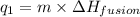
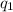 = amount of heat absorbed = ?
= amount of heat absorbed = ?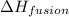 = enthalpy change for fusion = 127.40 J/g
= enthalpy change for fusion = 127.40 J/g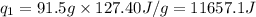
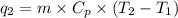
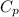 = specific heat capacity of benzene = 1.73 J/g°C
= specific heat capacity of benzene = 1.73 J/g°C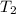 = final temperature = 60.6°C
= final temperature = 60.6°C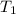 = initial temperature = 5.5°C
= initial temperature = 5.5°C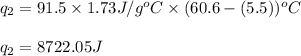
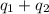
![[11657.1+8722.05]J=20379.2=20.38kJ](/tpl/images/0532/2114/b62c3.png)





