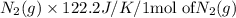Chemistry, 03.03.2020 06:05, Frenchfries13
Consider the reaction: N2(g) + 2O2(g)2NO2(g) Using standard absolute entropies at 298K, calculate the entropy change for the system when 1.90 moles of N2(g) react at standard conditions. S°system = J/K Submit Answer

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:10, bettybales1986
What effect does nuclear radiation have on atoms?
Answers: 1


Chemistry, 22.06.2019 10:50, mi364
A100 kmol/h stream that is 97 mole% carbon tetrachloride (ccl4) and 3% carbon disulfide (cs2) is to be recovered from the bottom of a distillation column. the feed to the column is 16 mole% cs2 and 84% ccl4, and 2% of the ccl4 entering the column is contained in the overhead stream leaving the top of the column. calculate the mass and mole fractions of ccl4 in the overhead stream, and determine the molar flow rates of ccl4 and cs2 in the overhead and feed streams. 12. mw_ccla- 153.82; mw_cs2-76.14.
Answers: 3

Chemistry, 22.06.2019 19:00, hmontalvo22
How many moles are contained in 5.6 l of h2 at stp
Answers: 3
Do you know the correct answer?
Consider the reaction: N2(g) + 2O2(g)2NO2(g) Using standard absolute entropies at 298K, calculate th...
Questions in other subjects:

History, 12.02.2021 04:10



Mathematics, 12.02.2021 04:10

Physics, 12.02.2021 04:10


Mathematics, 12.02.2021 04:10

English, 12.02.2021 04:10

Mathematics, 12.02.2021 04:10

Mathematics, 12.02.2021 04:10


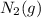 is 191.6 J/mol K,
is 191.6 J/mol K, 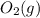 = 205 J/mol K, and
= 205 J/mol K, and 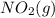 is 239.7 J/mol K at 298 K.
is 239.7 J/mol K at 298 K.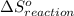 from standard absolute entropies as follows.
from standard absolute entropies as follows.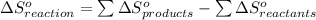
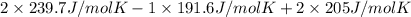
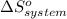 = 1.90 moles of
= 1.90 moles of