
Write the balanced net ionic equation for the reactions that occur when the given aqueous solutions are mixed. Include the physical states. A. silver nitrate, AgNO 3 AgNO3 , and magnesium bromide, MgBr 2 MgBr2 net ionic equation: B. perchloric acid, HClO 4 HClO4 , and potassium hydroxide, KOH KOH net ionic equation: C. ammonium sulfide, ( NH 4 ) 2 S (NH4)2S , and cobalt(II) chloride, CoCl 2 CoCl2 net ionic equation:

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 23:30, lizdeleon248
The sum of the oxidation numbers in a neutral compound is always
Answers: 2

Do you know the correct answer?
Write the balanced net ionic equation for the reactions that occur when the given aqueous solutions...
Questions in other subjects:

English, 17.01.2020 23:31




Chemistry, 17.01.2020 23:31

History, 17.01.2020 23:31




Social Studies, 17.01.2020 23:31


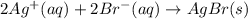
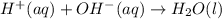
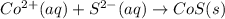
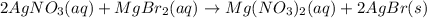
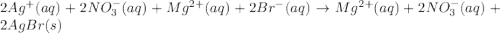
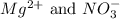 are the spectator ions.
are the spectator ions.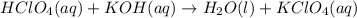
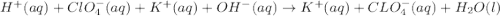
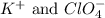 are the spectator ions.
are the spectator ions.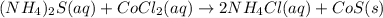
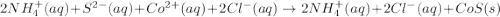
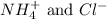 are the spectator ions.
are the spectator ions.



