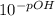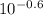A chemist must prepare 0.9 L of sodium hydroxide solution with a pH of 13.40 at 25°C. He will do this in three steps: Fill a 0.9 L volumetric flask about halfway with distilled water. Weigh out a small amount of solid sodium hydroxide and add it to the flask. Fill the flask to the mark with distilled water. Calculate the mass of sodium hydroxide that the chemist must weigh out in the second step.

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 00:00, familyk0jj3
How do you determine the percent yield of a chemical reaction
Answers: 1
Do you know the correct answer?
A chemist must prepare 0.9 L of sodium hydroxide solution with a pH of 13.40 at 25°C. He will do thi...
Questions in other subjects:

Mathematics, 20.05.2021 17:00





Mathematics, 20.05.2021 17:00

Mathematics, 20.05.2021 17:00