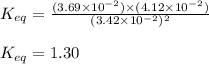Chemistry, 03.03.2020 03:22, icantspeakengles
Some CH2Cl2 is placed in a sealed flask and heated to 517 K. When equilibrium is reached, the flask is found to contain CH2Cl2 (3.42×10-2 M), CH4 (3.69×10-2 M), and CCl4 (4.12×10-2 M). What is the value of the equilibrium constant for this reaction at 517 K?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, Kaylinne1181
This chart represents the melting point of several substance. what besy explains the high melting point of the salt?
Answers: 2

Chemistry, 22.06.2019 09:40, gonzaleze18
In the lab, ammonia was mixed with water to form ammonium hydroxide. what is/are the reactant(s)? o water and ammonia o ammonia o ammonium hydroxide need
Answers: 2


Chemistry, 22.06.2019 13:00, torigirl4126
In what environment would mineral formation caused by high pressures and high temperatures most likely occur?
Answers: 3
Do you know the correct answer?
Some CH2Cl2 is placed in a sealed flask and heated to 517 K. When equilibrium is reached, the flask...
Questions in other subjects:

History, 19.11.2020 19:40

English, 19.11.2020 19:40


World Languages, 19.11.2020 19:40

Mathematics, 19.11.2020 19:40


Mathematics, 19.11.2020 19:40

English, 19.11.2020 19:40



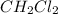 follows:
follows: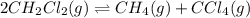
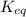 for above equation follows:
for above equation follows:![K_{eq}=\frac{[CH_4][CCl_4]}{[CH_2Cl_2]^2}](/tpl/images/0531/6381/cdd40.png)
![[CH_4]_{eq}=3.69\times 10^{-2}M](/tpl/images/0531/6381/50f3b.png)
![[CCl_4]_{eq}=4.12\times 10^{-2}M](/tpl/images/0531/6381/3e13d.png)
![[CH_2Cl_2]_{eq}=3.42\times 10^{-2}M](/tpl/images/0531/6381/c3f41.png)