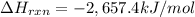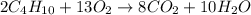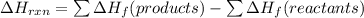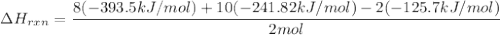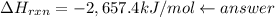Butane C4 H10 (g),(Delta. Hf = –125.7), combusts in the presence of oxygen to form CO2 (g) (Delta. Hf = –393.5 kJ/mol), and H2 O(g) (Delta. Hf = –241.82) in the reaction:
2 upper C subscript 4 upper H subscript 10 (g) plus 13 upper O subscript 2 (g) right arrow 8 upper C upper O subscript 2 plus 10 upper H subscript 2 upper O (g).
What is the enthalpy of combustion, per mole, of butane?
Use Delta H r x n equals the sum of delta H f of all the products minus the sum of delta H f of all the reactants..
–5,314.8 kJ/mol
–2,657.4 kJ/mol
2,657.4 kJ/mol
5,314.8 kJ/mol

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:40, alexandraparava
For a patient with the following pes statement and interventions, which would be the most appropriate monitoring and evaluating data? pes statement: inadequate calcium intake related to food and nutrition related knowledge deficit as evidenced by statements that the only dietary source of calcium is milk and she believes that she is lactose intolerant. patient’s nutrition prescription is for a diet providing 1200 mg calcium per day. patient was provided with in-depth nutrition education on alternative dietary and supplement sources of calcium. a. calcium intake (at subsequent visit) b. knowledge assessment by asking patient to identify food sources from menus and shopping list (at the end of the current visit) c. serum calcium (at next visit) d. both a and b e. both a and c
Answers: 2

Chemistry, 22.06.2019 04:30, anthony4034
Use the periodic table to determine the electron configuration of dysprosium (dy) and americium (am) in noble-gas notation.
Answers: 1

Chemistry, 22.06.2019 10:10, dhailyortegacampa131
Stage in which a typical star has completely stopped fusion
Answers: 1

Chemistry, 22.06.2019 13:30, citlalli30
1) which of the following is the best example of a physical change? a) sugar dissolving in tea b) firefly glowing 2) in the combustion of ethane, what is/are the reactants? c2h6 + o2 ==> co2 + h2o a) c2h6 and o2 b) co2 and c2h6
Answers: 2
Do you know the correct answer?
Butane C4 H10 (g),(Delta. Hf = –125.7), combusts in the presence of oxygen to form CO2 (g) (Delta. H...
Questions in other subjects:

Biology, 02.07.2019 20:30

English, 02.07.2019 20:30


Mathematics, 02.07.2019 20:30






Mathematics, 02.07.2019 20:30