
Chemistry, 02.03.2020 21:50, youngchapo813p8d9u1
If the pH of a somewhat dilute (i. e. ~0.08 to 0.10 M) aqueous solution of HCl is 1.14, then to a good approximation, what is the concentration of hydrogen ions in this solution? Enter your answer to the nearest hundredth.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:30, andrejr0330jr
What is the molar mass of potassium nitrate, kno3
Answers: 1

Chemistry, 22.06.2019 18:00, kingamir
Answer asap need it by wednesday morning carry out the following calculations on ph and ka of from data. i. calculate the ph of 0.02m hcl ii. calculate the ph of 0.036m naoh iii. calculate the ph of 0.36m ca(oh)2 iv. calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 v. calculate ka for weak acid ha which has a ph of 3.65 at 0.30m concentration vi. calculate the ka of a solution made by mixing 15.0 cm3 0.2m ha and 60.0 cm3 0.31m a-. [ph= 3.80] vii. calculate the ph of a solution made by mixing 15.0 cm3 0.1m naoh and 35.0 cm3 0.2m hcooh. [ka = 1.82 x 10-4 m]
Answers: 1

Chemistry, 22.06.2019 20:00, teacherpreacher
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2

Chemistry, 22.06.2019 22:00, notearslefttocry14
Imagine one batch of soup (batch “a”) is made with 8.19 g/can of salt, according to the recipe, and a second batch of soup (batch “b”) is made with 8.32 g/can of salt. explain which batch would be more resistant to frost damage if it is shipped a great distance in winter and explain why.
Answers: 2
Do you know the correct answer?
If the pH of a somewhat dilute (i. e. ~0.08 to 0.10 M) aqueous solution of HCl is 1.14, then to a go...
Questions in other subjects:


Physics, 14.02.2022 14:00

Physics, 14.02.2022 14:00

English, 14.02.2022 14:00


Mathematics, 14.02.2022 14:00


Mathematics, 14.02.2022 14:00


Health, 14.02.2022 14:00


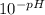 =
= 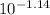 = 0.0724 M= 0.07 M
= 0.0724 M= 0.07 M



