Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, 767sebmont
Temperature and kinetic energy are proportional. a) adirectly b) directly c) indirectly
Answers: 2


Do you know the correct answer?
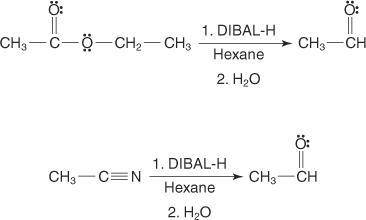
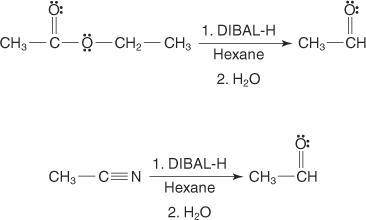
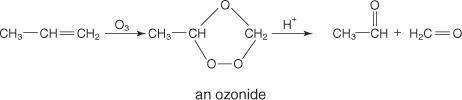
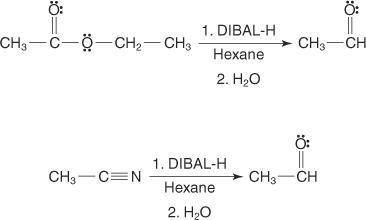
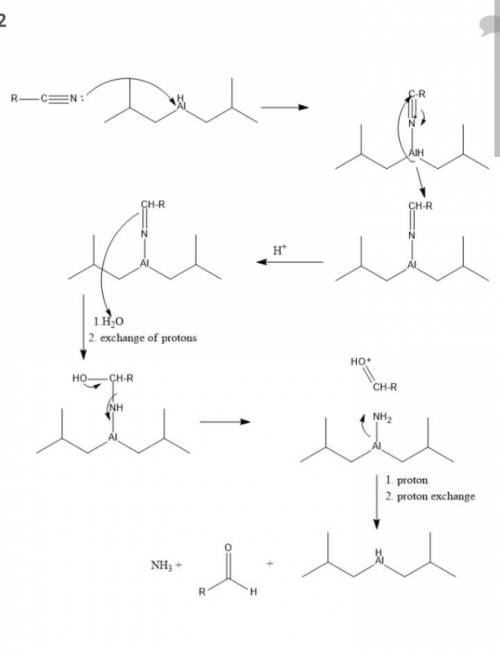
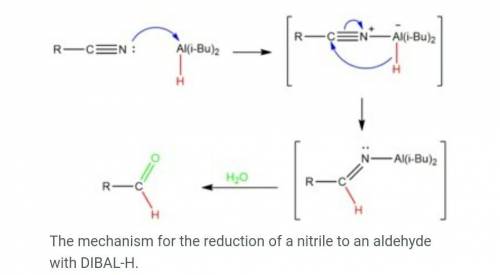
The reagent diisobutylaluminum hydride (DIBALH) reduces esters to aldehydes. When nitriles are treat...
Questions in other subjects:



Social Studies, 09.09.2020 19:01

Mathematics, 09.09.2020 19:01


Computers and Technology, 09.09.2020 19:01

Biology, 09.09.2020 19:01