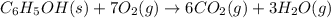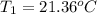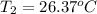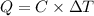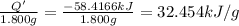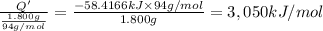A 1.800-g sample of solid phenol (C6H5OH(s)) was burned in a bomb calorimeter whose total heat capacity is 11.66 kJ/?C. The temperature of the calorimeter plus contents increased from 21.36?Cto 26.37?C. Part A
Write a balanced chemical equation for the bomb calorimeter reaction.
Part B:
What is the heat of combustion per gram of phenol?Part C:
Per mole of phenol?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:50, ttangelique
What are the 4 phases of matter in order of increasing engery content?
Answers: 2

Chemistry, 22.06.2019 09:30, junkmailemail42
Which element is the least metallic between cadmium, silver, zinc, or iron?
Answers: 1

Chemistry, 22.06.2019 12:00, winterblanco
What is the lowest number energy level where a d sublevel is found
Answers: 1

Chemistry, 22.06.2019 14:00, leahstubbs
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3
Do you know the correct answer?
A 1.800-g sample of solid phenol (C6H5OH(s)) was burned in a bomb calorimeter whose total heat capac...
Questions in other subjects:


Mathematics, 15.07.2019 03:30

Chemistry, 15.07.2019 03:30



Mathematics, 15.07.2019 03:30



Mathematics, 15.07.2019 03:30

Biology, 15.07.2019 03:30