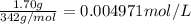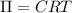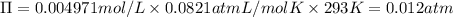Chemistry, 02.03.2020 16:58, gjeaneasley
Wastewater discharged into a stream by a sugar refinery contains 1.70 g of sucrose (C12H22O11) per liter. A government-industry project is designed to test the feasibility of removing the sugar by reverse osmosis. What pressure must be applied to the apparatus at 20.°C to produce pure water?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 23.06.2019 00:40, joe7977
To prevent the presence of air, noble gases are placed over highly reactive chemicals to act as inert "blanketing" gases. a chemical engineer places a mixture of noble gases consisting of 4.37 g of he, 13.36 g of ne, and 36.65 g of kr in a piston-cylinder assembly at stp. calculate the partial pressure in torr of kr.
Answers: 1

Chemistry, 23.06.2019 02:30, roseemariehunter12
Asubstance is held in an open container. its particles move past one another at random speeds but do not leave the container. heat is removed from the system, and the particles slow down. when enough heat is removed, the particles no longer have enough speed to overcome the weak attractive forces between them. when this happens, the substance enters its solid state. the process described above is known as .
Answers: 3

Chemistry, 23.06.2019 09:10, amandapill
In a 28 g serving of cheese curls there are 247mg of sodium. how much sodium is in a 12.5 ounce bag
Answers: 1
Do you know the correct answer?
Wastewater discharged into a stream by a sugar refinery contains 1.70 g of sucrose (C12H22O11) per l...
Questions in other subjects:


Mathematics, 12.02.2021 01:40

Mathematics, 12.02.2021 01:40

Mathematics, 12.02.2021 01:40

Social Studies, 12.02.2021 01:40

Health, 12.02.2021 01:40


Mathematics, 12.02.2021 01:40

Mathematics, 12.02.2021 01:40

Chemistry, 12.02.2021 01:40