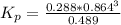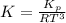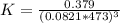Chemistry, 02.03.2020 16:54, cristinanina
Consider the decomposition of the compound C5H6O3 as follows below. C5H6O3(g) → C2H6(g) + 3 CO(g) When a 5.63-g sample of pure C5H6O3(g) was sealed into an otherwise empty 2.50 L flask and heated to 200.°C, the pressure in the flask gradually rose to 1.63 atm and remained at that value. Calculate K for this reaction.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:20, nyceastcoast
Give the orbital configuration of the phosphorus (p) atom.
Answers: 1


Chemistry, 22.06.2019 16:40, westball101
Let the ed50 of a recreational drug be defined as the amount required for 50% of a test group to feel high or get a buzz. if the ed50 value of ethanol is 470 mg/kg body mass, what dose would a 70 kg party goer need to quickly consume in order to have a 50% chance of getting a buzz? 235 mg 470 mg 32,900 mg 35,000,000 mg
Answers: 3

Chemistry, 23.06.2019 09:00, ashhull2002
Need ! assume that the variables x and y are directly related. if k = 8, what is the value for each of the following points? be sure and record your data to be used in the following problem. x y k 0.
Answers: 2
Do you know the correct answer?
Consider the decomposition of the compound C5H6O3 as follows below. C5H6O3(g) → C2H6(g) + 3 CO(g) Wh...
Questions in other subjects:

Mathematics, 05.07.2020 19:01

Biology, 05.07.2020 19:01

Chemistry, 05.07.2020 19:01


Mathematics, 05.07.2020 19:01

Mathematics, 05.07.2020 19:01


Mathematics, 05.07.2020 19:01


Mathematics, 05.07.2020 19:01