
Chemistry, 02.03.2020 17:06, aariannahnorwoo
At a particular temperature, K = 4.1 ✕ 10−6 for the following reaction. 2 CO2(g) 2 CO(g) + O2(g) If 2.3 moles of CO2 is initially placed into a 4.9-L vessel, calculate the equilibrium concentrations of all species.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:00, Killion2022
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1

Chemistry, 23.06.2019 00:00, scottykinkade7860
Mercury turns to a vapor at 629.88 k. how much heat is lost when 75.0 g of mercury vapor at 650 k condenses to a liquid at 297 k?
Answers: 1

Chemistry, 23.06.2019 02:00, xbeatdroperzx
Which statement is true about the model of the electromagnetic spectrum a: it change the frequencies of light. b: it compare wavelengths of light. c: the color of light waves can be changed using the model. d: the intensities of light waves can be decreased using the model.
Answers: 2
Do you know the correct answer?
At a particular temperature, K = 4.1 ✕ 10−6 for the following reaction. 2 CO2(g) 2 CO(g) + O2(g) If...
Questions in other subjects:




Mathematics, 26.01.2021 22:00

Mathematics, 26.01.2021 22:00

History, 26.01.2021 22:00




Mathematics, 26.01.2021 22:00


![[O_2]](/tpl/images/0530/1146/b0db0.png) = 0.0124 = 12.4 ×10⁻³ M
= 0.0124 = 12.4 ×10⁻³ M![[CO]](/tpl/images/0530/1146/32558.png) = 0.0248 = 2.48 ×10⁻² M
= 0.0248 = 2.48 ×10⁻² M![[CO_2]](/tpl/images/0530/1146/5494d.png) = 0.4442 M
= 0.4442 M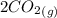 ⇄
⇄ 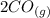 +
+ 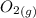
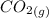 =
= 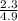 = 0.469
= 0.469![\frac{[CO]^2[O]}{[CO_2]^2}](/tpl/images/0530/1146/9940b.png)
![\frac{[2x]^2[x]}{[0.469-2x]^2}](/tpl/images/0530/1146/25de6.png)
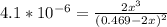
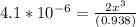
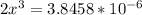
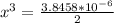
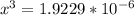
![x=\sqrt[3]{1.9929*10^{-6}}](/tpl/images/0530/1146/6ad64.png)





