
Chemistry, 02.03.2020 17:10, samstuart10p3g96w
Determine the value of the equilibrium constant, Kgoal, for the reaction N2(g)+O2(g)+H2(g)⇌12N2H4(g)+NO2(g), Kgoal=? by making use of the following information: 1. N2(g)+O2(g)⇌2NO(g), K1 = 4.10×10−31 2. N2(g)+2H2(g)⇌N2H4(g), K2 = 7.40×10−26 3. 2NO(g)+O2(g)⇌2NO2(g), K3 = 6.00×10−13 Express your answer numerically.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:50, hadwell34
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 22.06.2019 14:50, alexabbarker9781
How are evaporation and sublimation similar? a both involve the formation of a gas. b both release energy to the surroundings. c both take place throughout a solid. d both take place at the surface of a liquid.
Answers: 1
Do you know the correct answer?
Determine the value of the equilibrium constant, Kgoal, for the reaction N2(g)+O2(g)+H2(g)⇌12N2H4(g)...
Questions in other subjects:

Mathematics, 18.02.2021 18:40

Mathematics, 18.02.2021 18:40


Mathematics, 18.02.2021 18:40


English, 18.02.2021 18:40

Mathematics, 18.02.2021 18:40

Mathematics, 18.02.2021 18:40

English, 18.02.2021 18:40


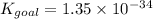
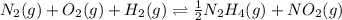
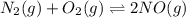 ;
; 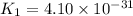
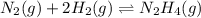 ;
; 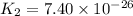
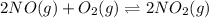 ;
; 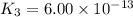
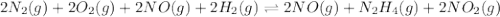
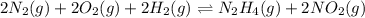 ...........(1)
...........(1)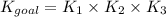
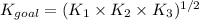
![K_{goal}=[(4.10\times 10^{-31})\times (7.40\times 10^{-26})\times (6.00\times 10^{-13})]^{1/2}](/tpl/images/0530/1334/2f97d.png)





