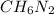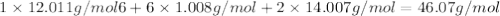Chemistry, 02.03.2020 07:15, snowprincess99447
Determine the molecular formula for the compound with a molar mass of 46.08 g/mol and the following percent compostion:
C:H:N:26.06%13.13%60.81%

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 21.06.2019 22:30, kkruvc
Will mark brainliest 26. which of these statements are true? (3 points) a. gases are compressible b. gases fill their containers completely c. the pressure of a gas is independent of the temperature d. gases have mass e. gases exert pressure f. the pressure of a gas is dependent on the volume g. gas pressure results from the collisions between gas particles h. gases have a definite volume and shape
Answers: 1


Chemistry, 22.06.2019 16:00, hjgjlgkjg
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1
Do you know the correct answer?
Determine the molecular formula for the compound with a molar mass of 46.08 g/mol and the following...
Questions in other subjects:

Mathematics, 27.05.2021 19:20



History, 27.05.2021 19:20

Mathematics, 27.05.2021 19:20

English, 27.05.2021 19:20

Mathematics, 27.05.2021 19:20