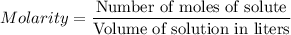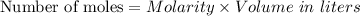Chemistry, 29.02.2020 01:25, samanthacruzsc51
For each trial, compute the mol of titrant; (molarity x L) and keep the number of significant figures to 4.
Trial 1: 12.49 mL =
mol NaOH
Trial 2: 12.32 mL =
mol NaOH
Trial 3: 11.87 mL =
mol NaOH

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:30, xoxokaydavis5837
You have 125g of a certain seasoning and are told that it contains 76.0 g of salt what is the percentage of salt by mass in this seasoning
Answers: 1


Chemistry, 23.06.2019 00:30, cashkidd2200
There are approximately 15 milliliters (ml) in 1 tablespoon (tbsp). what expression can be used to find the approximate number of milliliters in 3 tbsp?
Answers: 1

Chemistry, 23.06.2019 00:30, ejuarez2020
When a beta particle is emitted, the mass number of the nucleus a. decreases by one b. increases by one c. remains the same d. decreases by two
Answers: 2
Do you know the correct answer?
For each trial, compute the mol of titrant; (molarity x L) and keep the number of significant figure...
Questions in other subjects:

Mathematics, 19.01.2021 19:40


Mathematics, 19.01.2021 19:40

Biology, 19.01.2021 19:40


Health, 19.01.2021 19:40

English, 19.01.2021 19:40

Mathematics, 19.01.2021 19:40

Chemistry, 19.01.2021 19:40

English, 19.01.2021 19:40