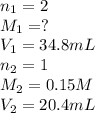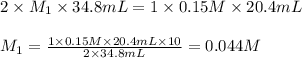Chemistry, 28.02.2020 23:18, smhrosepetals
If it takes 20.4 mL of NaOH(aq) to reach the equivalence point of the titration, what is the molarity of H2SO4(aq)? For your answer, only type in the numerical value with two significant figures. Do NOT include the unit.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:00, XxrazorxX11
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1

Chemistry, 22.06.2019 17:50, kaylamount
Cryolite, na3alf6(s), an ore used in the production of aluminum, can be synthesized using aluminum oxide. start this question by first balance the chemical equation.1.) balance the equation: - alo3(s)+naoh(l)+hf(> na3alf6+h2o(g). 2.) if 17.5 kilograms of al2o3(s), 51.4 kilograms of naoh(l), and 51.4 kilograms of hf(g) react completely, how many kilograms of cryolite will be produced? 3.)which reactants will be in excess, (al2o3, naoh, or hf) 4.)what is the total mass of the excess reactants left over after the reaction is complete in kg?
Answers: 2

Chemistry, 22.06.2019 20:30, dinapaul424
Which states of matter have particles that move independently of one another with very little attraction?
Answers: 1
Do you know the correct answer?
If it takes 20.4 mL of NaOH(aq) to reach the equivalence point of the titration, what is the molarit...
Questions in other subjects:

Mathematics, 06.05.2021 23:30

Chemistry, 06.05.2021 23:30


Mathematics, 06.05.2021 23:30


Mathematics, 06.05.2021 23:30


Mathematics, 06.05.2021 23:30



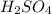 (aq) of an unknown concentration was titrated with 0.15 M of NaOH(aq).
(aq) of an unknown concentration was titrated with 0.15 M of NaOH(aq).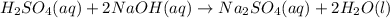
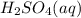 ? For your answer, only type in the numerical value with two significant figures. Do NOT include the unit.
? For your answer, only type in the numerical value with two significant figures. Do NOT include the unit.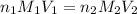
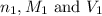 are the n-factor, molarity and volume of acid which is
are the n-factor, molarity and volume of acid which is 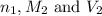 are the n-factor, molarity and volume of base which is NaOH.
are the n-factor, molarity and volume of base which is NaOH.