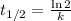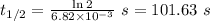Chemistry, 28.02.2020 19:45, brianadee800
The first-order rate constant for the decomposition of N2O5, 2N2O5(g)→4NO2(g)+O2(g) at 70∘C is 6.82×10−3 s−1. Starting with 8.00×10−2 mol of N2O5(g) in a volume of 2.9 L, how many moles of reactant are left after 5 minutes? What is its half-life?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, clairebear66
What three natural resources are found in the great lakes region
Answers: 2


Chemistry, 22.06.2019 23:00, jolainjoseph01998
What element has similar physical and chemical properties as boron.
Answers: 1

Chemistry, 23.06.2019 01:30, rubyr9975
What is the importance of interlocking the fingers and rubbing while washing hands? the palms are the dirtiest parts of the hands. the spaces between the fingers get washed. the backs of the hands get washed. the fingernails are the dirtiest parts of the hands
Answers: 1
Do you know the correct answer?
The first-order rate constant for the decomposition of N2O5, 2N2O5(g)→4NO2(g)+O2(g) at 70∘C is 6.82×...
Questions in other subjects:

Social Studies, 23.07.2019 03:30

Computers and Technology, 23.07.2019 03:30

English, 23.07.2019 03:30




Mathematics, 23.07.2019 03:30

Mathematics, 23.07.2019 03:30


History, 23.07.2019 03:30


![[A_t]=[A_0]e^{-kt}](/tpl/images/0528/2808/1ef89.png)
![[A_t]](/tpl/images/0528/2808/5262c.png) is the concentration at time t
is the concentration at time t ![[A_0]](/tpl/images/0528/2808/9a686.png) is the initial concentration =
is the initial concentration = 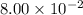 mol
mol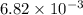 s⁻¹
s⁻¹![[A_t]=8.00\times 10^{-2}e^{-6.82\times 10^{-3}\times 300}\ mol=0.01034\ mol](/tpl/images/0528/2808/69780.png)