
Chemistry, 28.02.2020 19:28, alexantkoviak13
The rate of disappearance of HBr in the gas phase reaction 2HBr(g)→H2(g)+Br2(g) is 0.360 Ms−1 at 150 ∘C. The rate of appearance of Br2 is Ms−1. The rate of disappearance of in the gas phase reaction is 0.360 at 150 . The rate of appearance of is . 1.39 0.600 0.720 0.180 0.130

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:40, 4tazaouiamine1r
What type of solution is formed if 10 g of kclo3 are dissolved in 100g of water at 30
Answers: 2


Do you know the correct answer?
The rate of disappearance of HBr in the gas phase reaction 2HBr(g)→H2(g)+Br2(g) is 0.360 Ms−1 at 150...
Questions in other subjects:

Mathematics, 13.11.2020 05:40

Mathematics, 13.11.2020 05:40


Mathematics, 13.11.2020 05:40

Biology, 13.11.2020 05:40

Mathematics, 13.11.2020 05:40

English, 13.11.2020 05:40

Biology, 13.11.2020 05:40



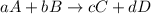
![\text{Rate of disappearance of A}=-\frac{1}{a}\frac{d[A]}{dt}](/tpl/images/0528/1964/2d8eb.png)
![\text{Rate of disappearance of B}=-\frac{1}{b}\frac{d[B]}{dt}](/tpl/images/0528/1964/1e77e.png)
![\text{Rate of formation of C}=+\frac{1}{c}\frac{d[C]}{dt}](/tpl/images/0528/1964/cee4b.png)
![\text{Rate of formation of D}=+\frac{1}{d}\frac{d[D]}{dt}](/tpl/images/0528/1964/7ef32.png)
![Rate=-\frac{1}{a}\frac{d[A]}{dt}=-\frac{1}{b}\frac{d[B]}{dt}=+\frac{1}{c}\frac{d[C]}{dt}=+\frac{1}{d}\frac{d[D]}{dt}](/tpl/images/0528/1964/d4b94.png)
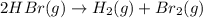
![\text{Rate of disappearance of }HBr=-\frac{1}{2}\frac{d[HBr]}{dt}](/tpl/images/0528/1964/d63dd.png)
![\text{Rate of appearance of }H_2=+\frac{d[H_2]}{dt}](/tpl/images/0528/1964/7fea8.png)
![\text{Rate of appearance of }Br_2=+\frac{d[Br_2]}{dt}](/tpl/images/0528/1964/d3d56.png)

![+\frac{d[Br_2]}{dt}=-\frac{1}{2}\frac{d[HBr]}{dt}](/tpl/images/0528/1964/3e9c6.png)
![\frac{d[Br_2]}{dt}=\frac{1}{2}\times 0.360M/s](/tpl/images/0528/1964/53e73.png)
![\frac{d[Br_2]}{dt}=0.180M/s](/tpl/images/0528/1964/3fdb1.png)





