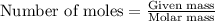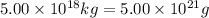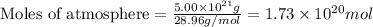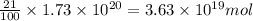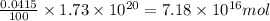The total mass of the atmosphere is about 5.00 x 1018 kg. How many moles each of air, O2, and CO2 are present in the atmosphere? Note that it is important to work in units of moles rather than in units of mass. By the ideal gas law, PV=nRT. P is pressure, V is volume, n is the number of moles, T is temperature (K), and R is the gas constant. At a given temperature and pressure, the volume is proportional to the number of moles, not to the mass.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:30, salvadorperez26
Suppose that during that icy hot lab 65,000 j of energy were transferred to 450 g of water at 20°c what would have have been the final temperature of the water
Answers: 2

Chemistry, 22.06.2019 05:30, medlinalex
Compare and contrast physical changes with chemical changes.
Answers: 1

Chemistry, 22.06.2019 15:00, hockeykid7583
Which are forms of frozen water? check all that apply. dew frost hail rain sleet
Answers: 2
Do you know the correct answer?
The total mass of the atmosphere is about 5.00 x 1018 kg. How many moles each of air, O2, and CO2 ar...
Questions in other subjects:


Mathematics, 22.01.2021 20:00


Computers and Technology, 22.01.2021 20:00



Mathematics, 22.01.2021 20:00

English, 22.01.2021 20:00

Chemistry, 22.01.2021 20:00

Mathematics, 22.01.2021 20:00


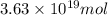 and
and 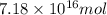 respectively
respectively