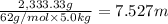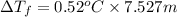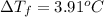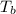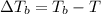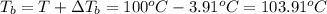Some ethylene glycol, , is added to your car’s cooling system along with 5.0 kg of water.
a. If the freezing point of the water–glycol solution is −14.0 °C, what mass of must have been added?
b. What is the boiling point of the coolant mixture? Kb(H20) = 0.52 degrees celcius kg mol^-1.

Answers: 2
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 20:00, bbyitskeke7160
What happens to the temperature of a substance when the average kinetic energy of its particles increases?
Answers: 3
Do you know the correct answer?
Some ethylene glycol, , is added to your car’s cooling system along with 5.0 kg of water.
Questions in other subjects:

Mathematics, 27.10.2020 21:10



Mathematics, 27.10.2020 21:10


History, 27.10.2020 21:10


Biology, 27.10.2020 21:10

Engineering, 27.10.2020 21:10


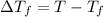
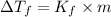
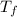 = freezing point of solution
= freezing point of solution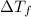 =depression in freezing point
=depression in freezing point 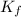 = freezing point constant
= freezing point constant 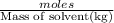
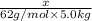
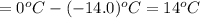
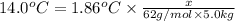
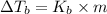
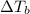 =elevation in boiling point =
=elevation in boiling point = 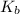 = boiling point constant
= boiling point constant