
Be sure to answer all parts. Consider the following equilibrium process at 686°C: CO2(g) + H2(g) ⇌ CO(g) + H2O(g) The equilibrium concentrations of the reacting species are [CO] = 0.0500 M, [H2] = 0.0410 M, [CO2] = 0.0880 M, and [H2O] = 0.0380 M. (a) Calculate Kc for the reaction at 686°C.(b) If we add CO2 to increase its concentrationto 0.30 mol/L, what will theconcentrations of all the gases be when equilibrium isreestablished?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 13:30, citlalli30
1) which of the following is the best example of a physical change? a) sugar dissolving in tea b) firefly glowing 2) in the combustion of ethane, what is/are the reactants? c2h6 + o2 ==> co2 + h2o a) c2h6 and o2 b) co2 and c2h6
Answers: 2

Chemistry, 22.06.2019 17:40, adantrujillo1234
Areaction in which products can react to re-form reactants is
Answers: 1

Chemistry, 22.06.2019 19:50, ellycleland16
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1
Do you know the correct answer?
Be sure to answer all parts. Consider the following equilibrium process at 686°C: CO2(g) + H2(g) ⇌ C...
Questions in other subjects:

Mathematics, 20.09.2019 08:00

Mathematics, 20.09.2019 08:00

Mathematics, 20.09.2019 08:00

Social Studies, 20.09.2019 08:00

Social Studies, 20.09.2019 08:00

Mathematics, 20.09.2019 08:00

Business, 20.09.2019 08:00


Chemistry, 20.09.2019 08:00

Biology, 20.09.2019 08:00


![[CO_2]=0.2834 M](/tpl/images/0526/9800/c4d54.png)
![[H_2]=0.02440 M](/tpl/images/0526/9800/4ccc4.png)
![[CO]=0.06660 M](/tpl/images/0526/9800/7fe11.png)
![[H_2O]=0.05460 M](/tpl/images/0526/9800/56dcb.png)
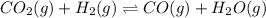
![[CO]=0.0500 M](/tpl/images/0526/9800/16f57.png)
![[H_2]=0.0410 M](/tpl/images/0526/9800/8d6da.png)
![[CO_2]=0.0880 M](/tpl/images/0526/9800/fdfdd.png)
![[H_2O]=0.0380 M](/tpl/images/0526/9800/4cb1c.png)
![K_c=\frac{[CO][H_2O]}{[CO_2][H_2]}](/tpl/images/0526/9800/c597d.png)
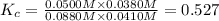

![[CO_2]=(0.30-x)=(0.30-0.0166) M=0.2834 M](/tpl/images/0526/9800/94c3e.png)
![[H_2]=(0.0410-x)=(0.0410-0.0166) M=0.02440 M](/tpl/images/0526/9800/e30dc.png)
![[CO]=(0.0500+x)=(0.0500+0.0166) M=0.06660 M](/tpl/images/0526/9800/af1c7.png)
![[H_2O]=(0.0380+x)=(0.0380+0.0166) M=0.05460 M](/tpl/images/0526/9800/b2417.png)




