
Chemistry, 27.02.2020 04:55, wileycheyennem
What is the solubility of calcium sulfate (CaSO4) in 0.30 M aqueous sodium sulfate (Na2SO4)? (Ksp of calcium sulfate = 2.0 x 10^-5

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, brittanysanders
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2

Chemistry, 22.06.2019 16:10, nauticatyson9
Given the following equation: 2a1 + 3mgcl2 --> 2alcl3 + 3mg how many moles of aluminum chloride are produced from 2.5 moles of magnesium chloride?
Answers: 1

Do you know the correct answer?
What is the solubility of calcium sulfate (CaSO4) in 0.30 M aqueous sodium sulfate (Na2SO4)? (Ksp of...
Questions in other subjects:

Advanced Placement (AP), 21.03.2021 22:30

Mathematics, 21.03.2021 22:30






Mathematics, 21.03.2021 22:30

Mathematics, 21.03.2021 22:30

Mathematics, 21.03.2021 22:30


![K_{sp}=[Ca^{2+}][SO_4^{2-}]](/tpl/images/0526/4512/958f2.png) ........(1)
........(1)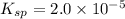
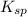 and [SO₄²⁻] in equation (1)
and [SO₄²⁻] in equation (1)![2.0\times 10^{-5}= [Ca^{2+}]0.30](/tpl/images/0526/4512/53779.png)
![\Rightarrow [Ca^{2+}]=\frac{2.0\times 10^{-5}}{0.30}](/tpl/images/0526/4512/70356.png)




